Iodine pentafluoride F5I CAS 7783-66-6
Get Latest Price| Payment Type: | L/C,T/T |
| Incoterm: | FOB,CFR |
| Min. Order: | 1 Kilogram |
| Transportation: | Ocean,Land,Air,Express |
| Port: | Shanghai Port,Qingdao Port,Ningbo Port |
Mr. zhaokui
What can I do for you?

| Payment Type: | L/C,T/T |
| Incoterm: | FOB,CFR |
| Min. Order: | 1 Kilogram |
| Transportation: | Ocean,Land,Air,Express |
| Port: | Shanghai Port,Qingdao Port,Ningbo Port |
Model No.: vsk 7783-66-6
Brand: Volksky
Classification: Other Inorganic Salts
Place Of Origin: China
Grade: Agriculture Grade, Electron Grade, Food Grade, Industrial Grade, Medicine Grade, Reagent Grade
| Selling Units | : | Kilogram |
| Package Type | : | 1kg bag,25kg drum; provide tailored package on request |
| Picture Example | : |

|
Iodine pentafluoride Basic information
Iodine pentafluoride (IF5) is a colorless to pale yellow fuming liquid with a pungent odor. The highly reactive IF5 (melting point 9.4 °C) is distillable at 100.5 °C without any decomposition. Decomposition commences at temperatures above 400 °C.
Iodine pentafluoride has good thermal stability and is the most stable halogen compound. The reactivity of IF5 is much weaker than that of other halogen compounds, and it does not interact with silicon, magnesium, copper, iron, and chromium at room temperature, and only interacts with molybdenum, tungsten, phosphorus, arsenic, antimony, boron, etc.
It is the least reactive of the halogen fluorides and classified as a soft fluorinating reagent. It is used as a mildest, selective fluorinating reagent in the fluorination of organic molecules.
Product Name: Iodine pentafluoride
Synonyms: Pentalfluoroiodide;Pentafluoroiodine;IF5;Iodine fluoride;Iodine fluoride (IF5);Iodinefluoride;iodinefluoride(if5);IODINE PENTAFLUORIDE
CAS: 7783-66-6
MF: F5I
MW: 221.9
Iodine pentafluoride Chemical Properties
| Melting point | 9.4°C |
| Boiling point | 104.5°C |
| density | 3,2 g/cm3 |
| vapor pressure | 26.664hPa at 20℃ |
| MF | 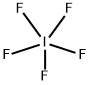 |
Ures
Iodine pentafluoride is a fluorinating agent and strong oxidant, which plays an important role in organic synthesis. It can be used to prepare pentafluoroiodoethane:
5 CF2=CF2 + IF5 + 2 I2 → 5 C2F5I
In addition, iodine pentafluoride can also react with isothiocyanato to generate fluorinated hydrocarbon derivatives, and to synthesize other fluorine-containing compounds.
Iodine pentafluoride (IF5) is rarely used to fluoride common organic compounds due to the difficulty in controlling their reactivity.


Privacy statement: Your privacy is very important to Us. Our company promises not to disclose your personal information to any external company with out your explicit permission.

Fill in more information so that we can get in touch with you faster
Privacy statement: Your privacy is very important to Us. Our company promises not to disclose your personal information to any external company with out your explicit permission.